Last Updated: May 03, 2026
Uploaded By: NotesLover
3 Views
19 Pages
Basic Concepts of Chemistry: Handwritten Notes PDF for NEET/JEE
Mastering the fundamentals is the first step toward cracking competitive exams like NEET and JEE. These Basic Chemistry Handwritten Notes are designed to simplify complex introductory topics, providing you with a solid foundation in Physical, Inorganic, and Organic chemistry.
Key Highlights of the PDF
- ✔ **Comprehensive Coverage**: From the definition of matter to complex unit conversions.
- ✔ **Memory Shortcuts**: Exclusive Hindi mnemonics (Tricks) for the Periodic Table.
- ✔ **Numerical Essentials**: Tables for atomic masses, molecular weights, and valencies[cite: 2].
- ✔ **Exam-Oriented**: Focus on scientific notation and the criss-cross method for formulas[cite: 2].
1. Introduction to the World of Chemistry
Chemistry is defined as the branch of science dealing with the composition, structure, and properties of matter[cite: 2]. Often referred to as the "Central Science," it explains how atoms interact to form the world around us. Antoine Laurent Lavoisier is recognized as the Father of Chemistry for his pioneering work[cite: 2].
The Three Pillars: Branches of Chemistry
According to the notes, chemistry is split into three main domains[cite: 2]:
- Physical Chemistry: Studies the behavior of matter at atomic and molecular levels[cite: 2].
- Inorganic Chemistry: Deals with elements and compounds excluding carbon-based structures[cite: 2].
- Organic Chemistry: Focuses specifically on carbon and its vast array of compounds[cite: 2].
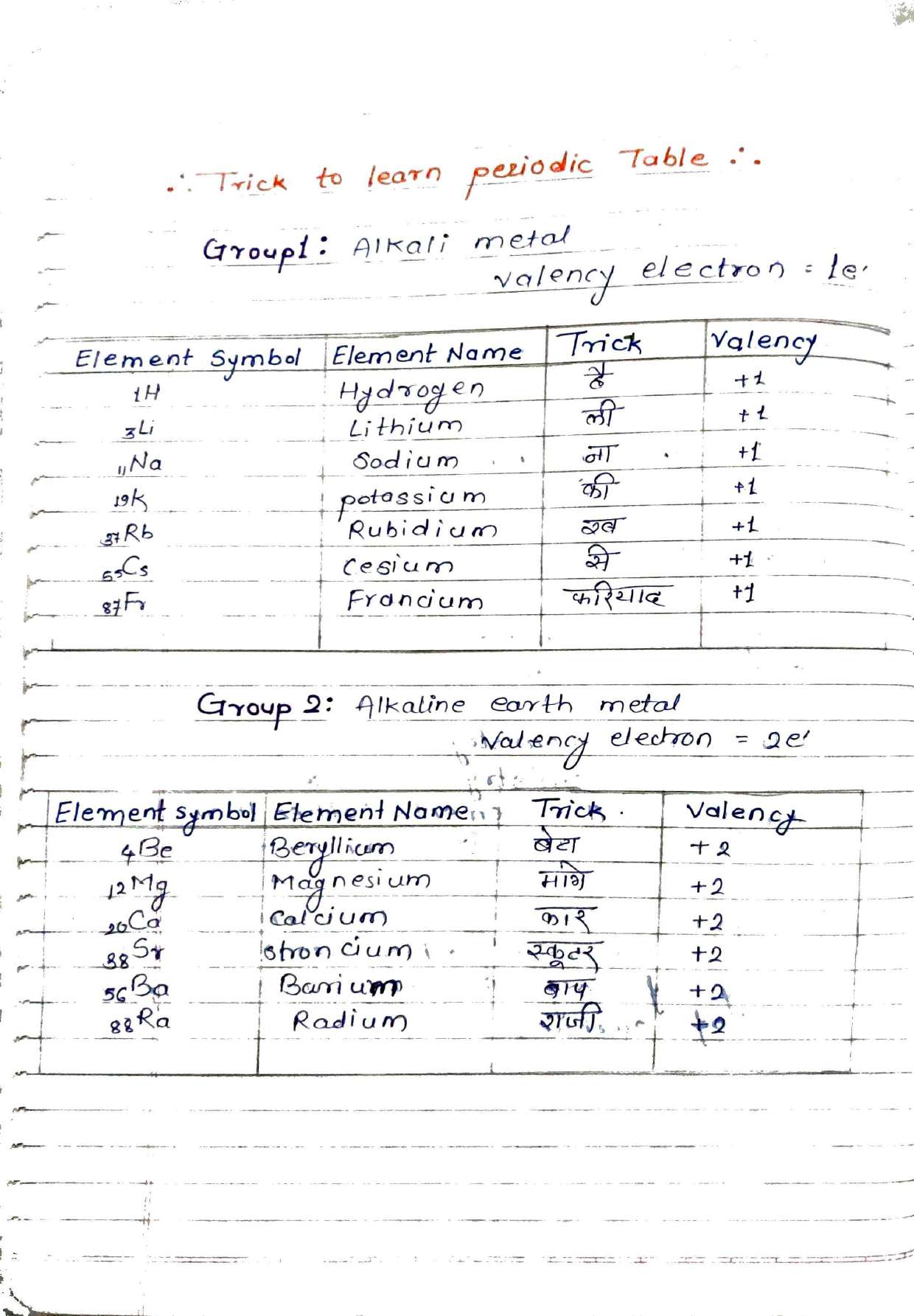
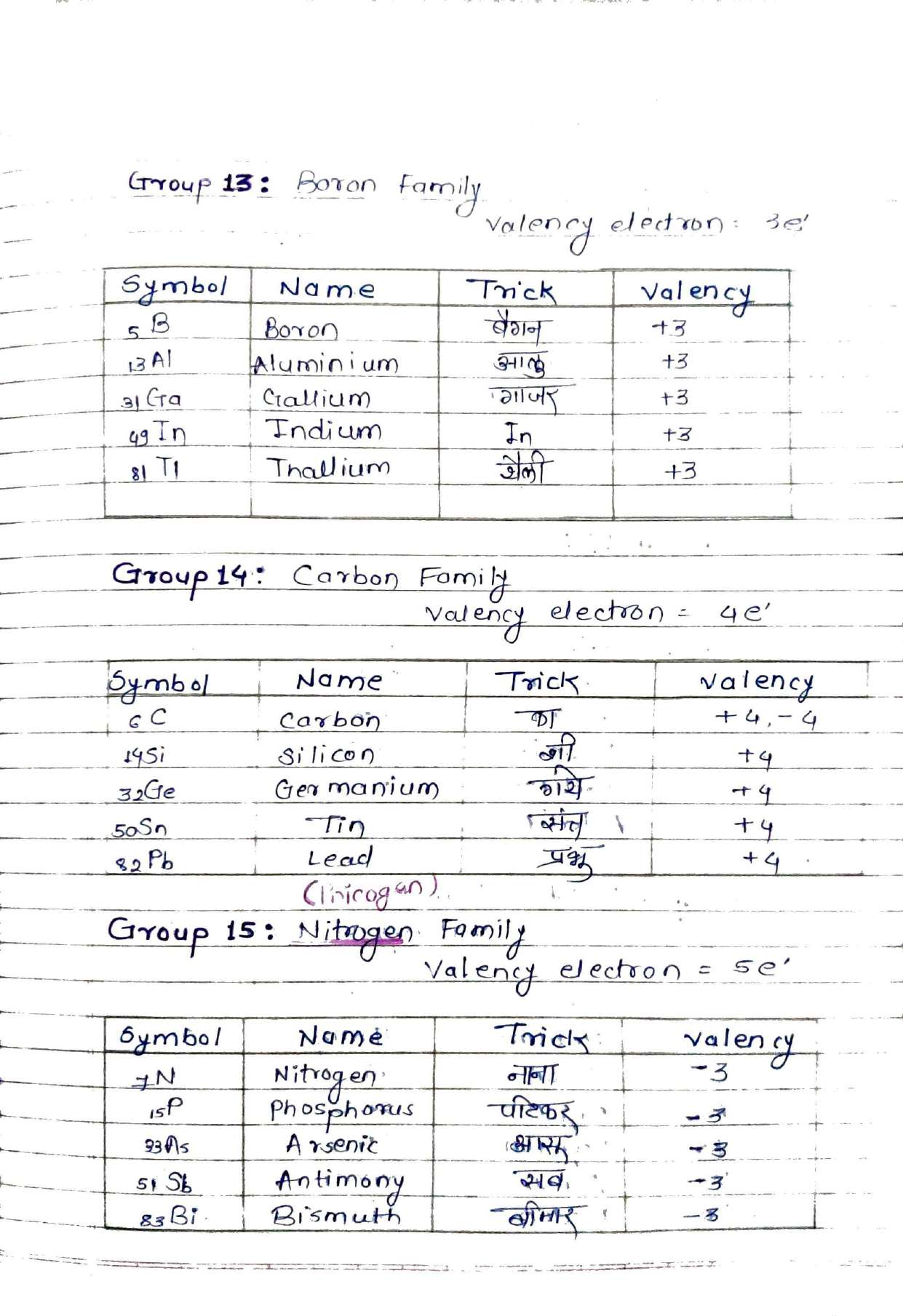
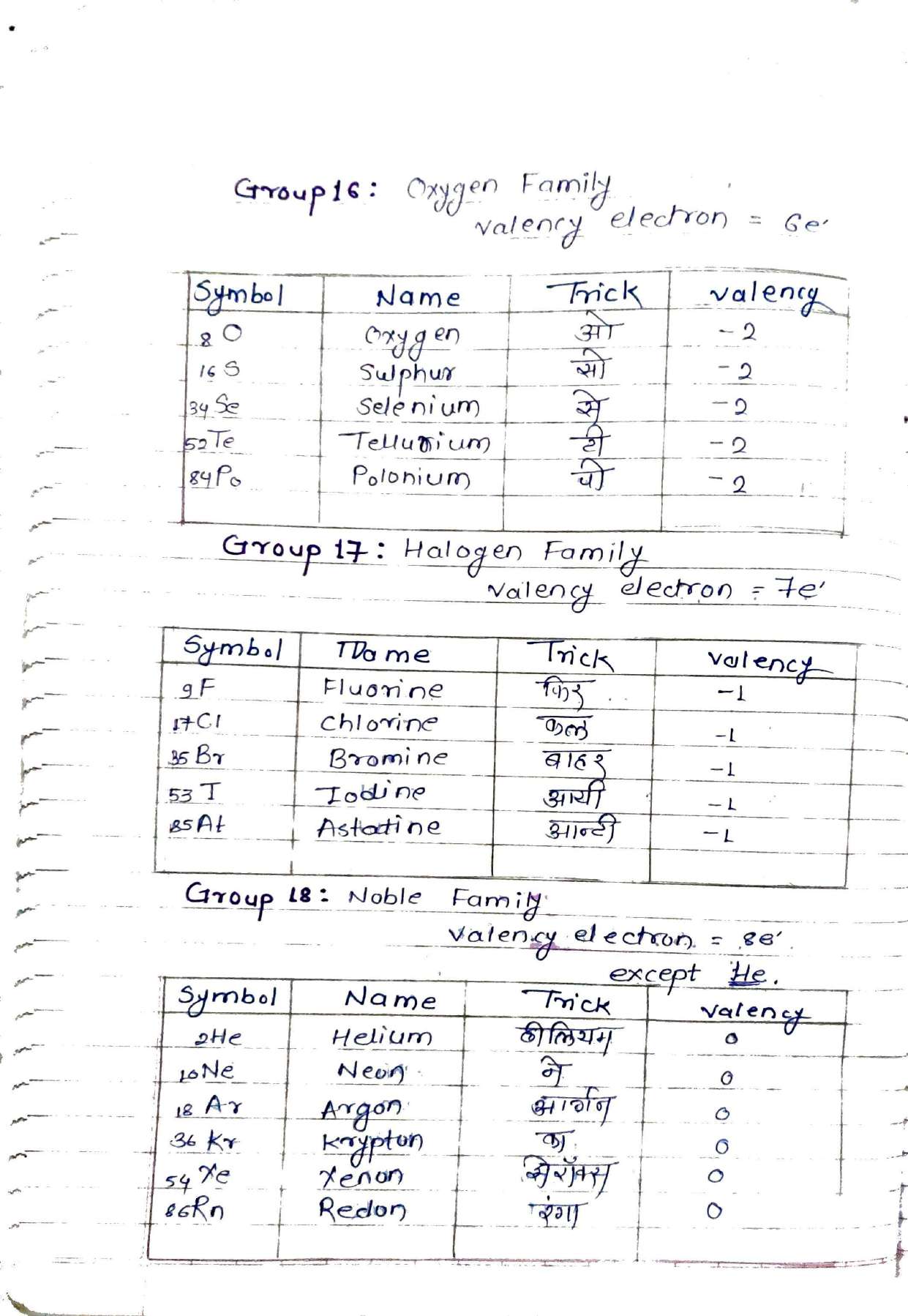
2. Periodic Table Tricks: "Ha-Li-Na-Ki-Rub-Se-Fariyad"
Memorizing 118 elements is daunting, but these notes provide unique mnemonics[cite: 2]. For example, the Alkali Metals (Group 1) can be remembered using the phrase "है ली ना की रब से फरियाद" (H, Li, Na, K, Rb, Cs, Fr)[cite: 2]. Similarly, the Halogen Family (Group 17) is simplified as "फिर कल बाहर आयी आन्टी" (F, Cl, Br, I, At)[cite: 2].
3. Atomic Representation and Calculations
An atom is the smallest part of an element participating in a chemical reaction[cite: 2]. It is represented as $zX^{A}$, where:
- Z: Atomic number (Number of protons)[cite: 2].
- A: Mass number (Protons + Neutrons)[cite: 2].
- Neutrons: Calculated as $A - Z$[cite: 2].
Mastering Molecular Mass
Calculating molecular mass is essential for stoichiometry. The PDF provides a step-by-step guide for common compounds[cite: 2]:
- Water ($H_2O$): $2(1) + 16 = 18$[cite: 2].
- Sulphuric Acid ($H_2SO_4$): $2(1) + 32 + 4(16) = 98$[cite: 2].
- Glucose ($C_6H_{12}O_6$): $12(6) + 1(12) + 16(6) = 180$[cite: 2].
- Urea ($NH_2CONH_2$): 60[cite: 2].
4. Valency and Polyatomic Radicals
Valency determines how atoms bond. The notes provide an extensive chart of ions[cite: 2]:
- Cations: $Na^{+1}, Mg^{+2}, Al^{+3}$[cite: 2].
- Anions: $Cl^{-1}, O^{-2}, N^{-3}$[cite: 2].
- Radicals: Sulphate ($SO_4^{2-}$), Nitrate ($NO_3^-$), and Phosphate ($PO_4^{3-}$)[cite: 2].
5. Scientific Notation & Unit Conversions
In Chemistry, we often deal with extreme numbers. Scientific notation represents these as $N \times 10^n$[cite: 2]. For instance, $0.00006$ becomes $6.0 \times 10^{-5}$[cite: 2].
Crucial Conversions for Numericals
Accurate unit conversion is the difference between a correct and incorrect answer in NEET[cite: 2]:
- Pressure: $1 \text{ atm} = 760 \text{ mmHg} = 760 \text{ torr}$[cite: 2].
- Temperature: $K = °C + 273.15$ and $°F = \frac{9}{5}°C + 32$[cite: 2].
- Energy: $1 \text{ calorie} = 4.18 \text{ Joules}$[cite: 2].
Frequently Asked Questions
Q1: Who is the father of Chemistry?
Antoine Laurent Lavoisier is called the father of chemistry[cite: 2].
Q2: What is the molecular mass of Potassium Dichromate?
The molecular mass of $K_2Cr_2O_7$ is calculated as 294[cite: 2].
Q3: How do you remember metalloids?
The PDF suggests the trick "आन्टी तेरी सहजी पूरी बासी बासी" to remember elements like At, Te, Sb, Ge, Po, B, As, and Si[cite: 2].
Q4: What are the main branches of chemistry?
Mainly three branches: Physical, Inorganic, and Organic chemistry[cite: 2].
Get the complete 26-page handwritten guide for your exam preparation.
DOWNLOAD FULL PDF NOTES(नोट्स नहीं मिल रहे? हम फ्री में देंगे!)
Disclaimer: NotesLover is an independent educational platform and is not affiliated with, endorsed by, or associated with any government body or recruitment board.
अस्वीकरण: NotesLover एक स्वतंत्र शैक्षिक मंच है और किसी भी सरकारी निकाय या भर्ती बोर्ड से संबद्ध, समर्थित या जुड़ा नहीं है।
NotesLover provides free educational notes for learning purposes only. Content owners may request removal. Click Here.
NotesLover केवल शैक्षणिक उद्देश्य के लिए निःशुल्क अध्ययन सामग्री प्रदान करता है। यदि आप सामग्री के स्वामी हैं और किसी भी सामग्री को हटवाना चाहते हैं, तो यहाँ क्लिक करें।
NEET Chemistry Chemistry Handwritten Notes Periodic Table Atomic Mass Physical Chemistry NEET 2026
Reviews
No review available.To leave a comment, please log in.
Log in to CommentFeatured Notes
Handwritten Notes on the Indian Constitution in Hindi (PDF)
Handwritten Linux Notes PDF | Simplified Linux Commands & Concepts for Beginners
Ancient Indian History: Chronological Study from Indus Valley to Gupta Empire
Comprehensive Reasoning Handwritten Notes PDF - SSC, RRB, Banking, Police, SI & Homeguard
Indian polity handwritten notes hindi | PDF
Modern Indian History Notes PDF
UP Board Class 10 Science Handwritten Notes in Hindi PDF (Latest Session)
UP Board Class 12 Physics Vol-2 Handwritten Notes PDF 2026
SSC GD & CGL Mathematics Handwritten Notes PDF - Percentage & Profit Loss (Half Chapter)
Complete SSC Maths Formula Guide for Competitive Exams
Ancient History Notes PDF - Free Download for All Competitive Exams
Light: Reflection and Refraction Class 10 Notes - PDF & Key Formulas
Related Notes
Ultimate NEET Physics Electromagnetism Notes: Comprehensive Handwritten Guide for 2026 Aspirants
The Ultimate NEET Physics Survival Guide: Master Mathematical Tools & Foundation Concepts
Complete NEET Zoology Handwritten Notes PDF - Human Reproduction & Biotechnology
NEET Chemistry Notes PDF Free Download
Animal Kingdom: Detailed Handwritten Notes PDF for Competitive Exams
© 2026 Notes Lover. All rights reserved.